

AEIC provides quality products and
systems to meet our solutions' requirement and satisfy our customers.
Fuel cell is newer energy source for building and industries; VRB-ESS is a
better energy storage for emergency power or UPS and CPS (continual power
supply system) up to few hours or the time you need.
To combine the fuel cell with
VRB-ESS, the new power generation system will not be outage forever.
AEIC
is a member of Singapore Fuel Cell Community.

![]() FUEL
CELL
FUEL
CELL
The fuel cell is an energy conversion device that can efficiently capture and use the power of hydrogen. Hydrogen is a versatile energy carrier that can be used by fuel cell as a fuel to power nearly every end-use energy need. Since the first fuel cell is invented, the fellow researchers have developed the materials and many types of fuel cells which have been applied in various industries, such as Proton Exchange Membrane Fuel Cell (PEMFC), Phosphoric Acid Fuel Cell (PAFC), Alkaline Fuel Cell, Molten Carbonate Fuel Cell (MCFC) and Solid Oxide Fuel Cell (SOFC). The comparison of various fuel cells is listed in below table.
Table – Comparison of various fuel cells (source from DOE)
The fuel cell relies on a basic oxidation / reduction reaction that takes place on the fuel rather than the electrodes. The fuel cell produces electricity as long as the cell receives fuel supply and can dispose of the oxidized fuel. In a fuel cell, the anode is usually bathed in the fuel; the cathode collects and makes the oxidant be available; an ion-conducting membrane separates the two of anode and cathode, but allows the reaction to take place without affecting the electrodes. The schematic of fuel cell is shown in below figure.
Figure –
Schematic of PEM Fuel Cell (source from DOE)
The fuel cell was forecasted shipments in 2011 surpassing 285,000 units worldwide with growing by 25% compared to 2010. The PEMFC leads 2011 unit shipments and accounts for over 95% of the total shipments, and North America regionally dominated with a 50% market share of total units. Those are mostly related to the material handling, combined heat and power (CHP), back-up power, and auxiliary power sectors in the global commercial market in 2010.
If recovering the heat completely from fuel cell, the fuel cell efficiency shall achieve 100% technically.
![]() VRB-ESS
VRB-ESS
Based on the technology, the Vanadium Redox (or redox flow) Battery (VRB) is a type of rechargeable flow battery that employs vanadium ions in different oxidation states to store chemical potential energy. VRB technology is a proven, economically attractive and low-maintenance solution for energy storage. When a group of VRBs are used for energy storage, the whole system becomes VRB-ESS.
VRB is an effective mean to accumulate electrical energy among the emerging technologies that the redox flow battery has. The flow batteries are excellent candidates for large stationary energy storage applications where the flow battery distinguishes itself thanks to its competitive cost and simplicity. VRB allows energy to be stored in two solutions containing different redox couples. Unlike conventional batteries, the redox flow cell stores energy in the solutions so that the capacity of the system is determined by the size of the electrolyte tanks, while the system power is determined by the size of the cell stacks. The below Figure demonstrates such a scheme with two cells in parallel.
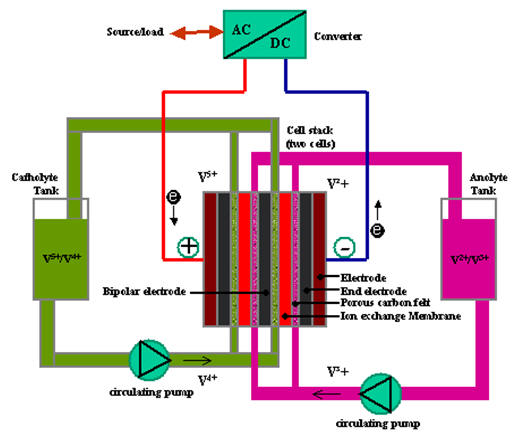
Figure – Principle Schematic of Vanadium Redox Battery
OPERATING PRINCIPLE
As the schematic shown in above Figure, a VRB has two chambers, named as positive chamber and negative chamber, separated by an ion exchange membrane and employs the V(III)/V(II) and V(V)/V(IV) redox couples in sulphuric acid as the negative and positive half-cell electrolytes respectively. During discharge process, V2+ is oxidized to V3+ at the negative electrode and VO2+ (V5+) is reduced to VO2+ (V4+) at the positive electrode, as shown in Equation (2.1), (2.2) and (2.3).
![]()
At the positive electrode:
VO2+ + 2H+ + e- <==========> VO2+ + H2O (E° = 1.00V via SHE) (2.1)![]()
At
the negative electrode:
V2+ - e- <==========> V3+ (E° = -0.26 V
via SHE)
(2.2)
![]()
Cell
reaction:
VO2+ + V2+ + 2H+ <==========> VO2+
+ V3+ + H2O (E° = 1.26 V via SHE)
(2.3)
This gives a standard cell potential E° (cell) = 1.26 Volts at concentrations of 1 mole per liter and at 25°C. Under actual cell conditions, an open-circuit cell voltage of 1.4 Volts is observed at 50% state-of-charge, while a fully charged cell produces over 1.6 Volts at open-circuit. The electrolyte for the VRB is 2 M vanadium sulphate in 2.5 M H2SO4, the vanadium sulphate [initially 1 M V (III) + 1 M V (IV)] being prepared by chemical reduction or electrolytic dissolution of V2O5 powder. The basic components of the redox cell are illustrated in below Figure.
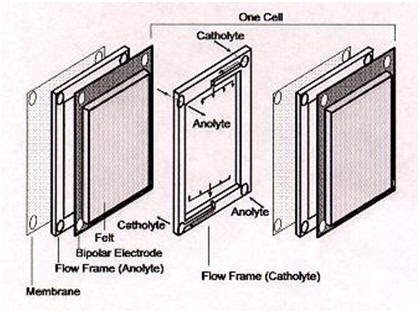
Figure – Components of the Redox Cell
During discharge process, the hydronium ions are transported across an ion-exchange membrane (IEM) from the negative electrode to the positive electrode. The reactions proceed in the opposite direction during charge process. The active species are normally dissolved in the sulphuric acid, and the protons transport across the ion-exchange membrane to balance the charge. The VRB uses two tanks to store electrolytes that are pumped through a battery stack where the chemical energy stored in the solutions is converted to electrical energy.
The VRB-ESS can be used for energy storage in small scale (kW/kWh) to large scale (MW/MWh) with smaller footprint. If VRB-ESS is integrated with power conditioning system (PCS), the system will become uninterrupted power system (UPS) up to tens MW/MWh and backup your essential systems as you need.
Contact us for detailed solution for your needs.
For more detailed information, please review our KQP's Paper - "Investigation of VRB-ESS integrated with hybrid power system for building" presented in the 10th International Power and Energy Conference (IPEC 2012) in Ho Chi Minh City, Vietnam and published in IEEE®
Sales Enquiries,
Technical Support at
[email protected]
Copyright 2003 - 2013 © www.aeics.com.
Best viewed with Internet Explorer 6.0, 800x600 resolution.